AJL VISC – AJL CELL
Viscoelastic Solutions
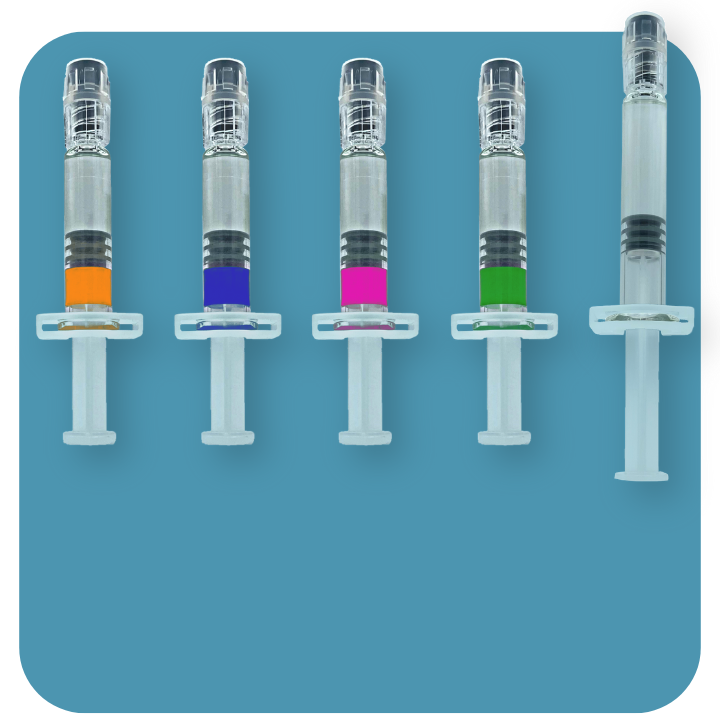
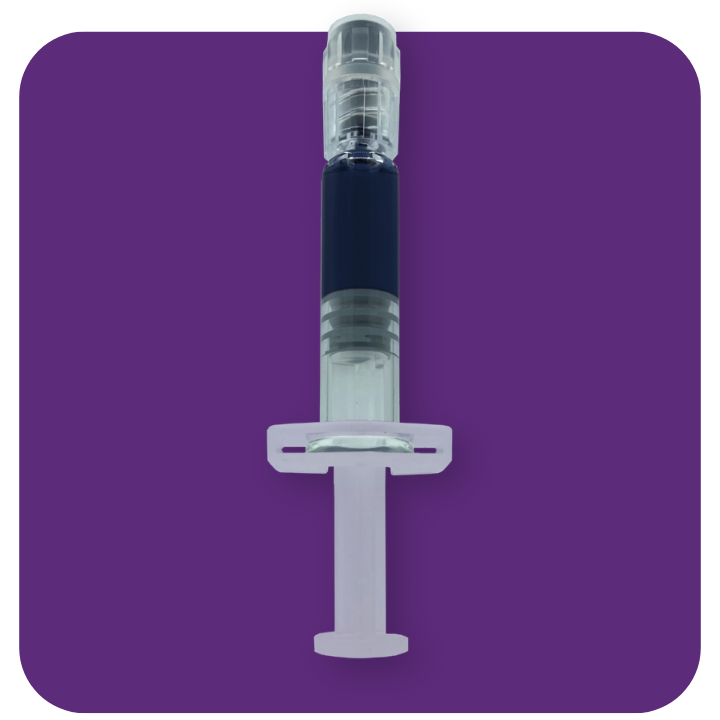
Non-absorbable viscoelastic solution, highly purified and non-inflammatory, isotonic, sterile and non-pyrogenic, for intraocular injection.

SPECS
| Brand name | |||||
| Reference | AJL VISC 1% | AJL VISC 1.4% | AJL VISC 2% | AJL VISC 3% | AJL CELL 2% |
| Description and indications | Non-absorbable viscoelastic solution, highly purified and non-inflammatory, isotonic, sterile and non-pyrogenic, for intraocular injection. The viscoelastic solutions are indicated as a surgical aid in surgical procedures in the anterior segment of the eye, including cataract surgery. AJL VISC and AJL CELL keeps the depth of the anterior chamber, providing a high protection of the tissues and avoiding the risk of corneal endothelium trauma, as weel as other ocular tissues. |
||||
| Properties | • High endothelium protection. • Excellent rheological properties. • Excellent rheological properties. • Excellent visibility of the treated area. • Easy to remove from the anterior chamber. |
||||
| Composition / Molecular Weight (MW) | MW: 2.400.000 Da | MW: 92.000 Da |
|||
| Viscosity (1 S-1 25°C) | 10000-14000 mPa.s | 15000-20000 mPa.s | 40 000-60000 mPa.s | 160 000-200 000 mPa.s | 2 000-4 000 mPa.s |
| pH | 7.0-7.5 | 7.0-7.5 | 7.0-7.5 | 7.0-7.5 | 7.0-7.5 |
| Osmolarity | 250-350 mOsmol/l | 250-350 mOsmol/l | 250-350 mOsmol/l | 250-350 mOsmol/l | 250-350 mOsmol/l |
| Volume | 1 ml. | 1 ml. | 1 ml. | 1 ml. | 2 ml. |
| Supply | Sterile, single unit. Prefilled glass syringe of 1 ml. Luer lock 27G cannula. Transparent backstop. | Sterile, single unit. Prefilled glass syringe of 1 ml. Luer lock 27G cannula. Transparent backstop. | Sterile, single unit. Prefilled glass syringe of 1 ml. Luer lock 27G cannula. Transparent backstop. | Sterile, single unit. Prefilled glass syringe of 1 ml. Luer lock 25G cannula. Transparent backstop. | Sterile, single unit. Prefilled glass syringe of 2 ml. Luer lock 27G cannula. Transparent backstop. |
| Sterilization method | Steam. | ||||
| Shelf-life | 3 years | ||||
CE 2797